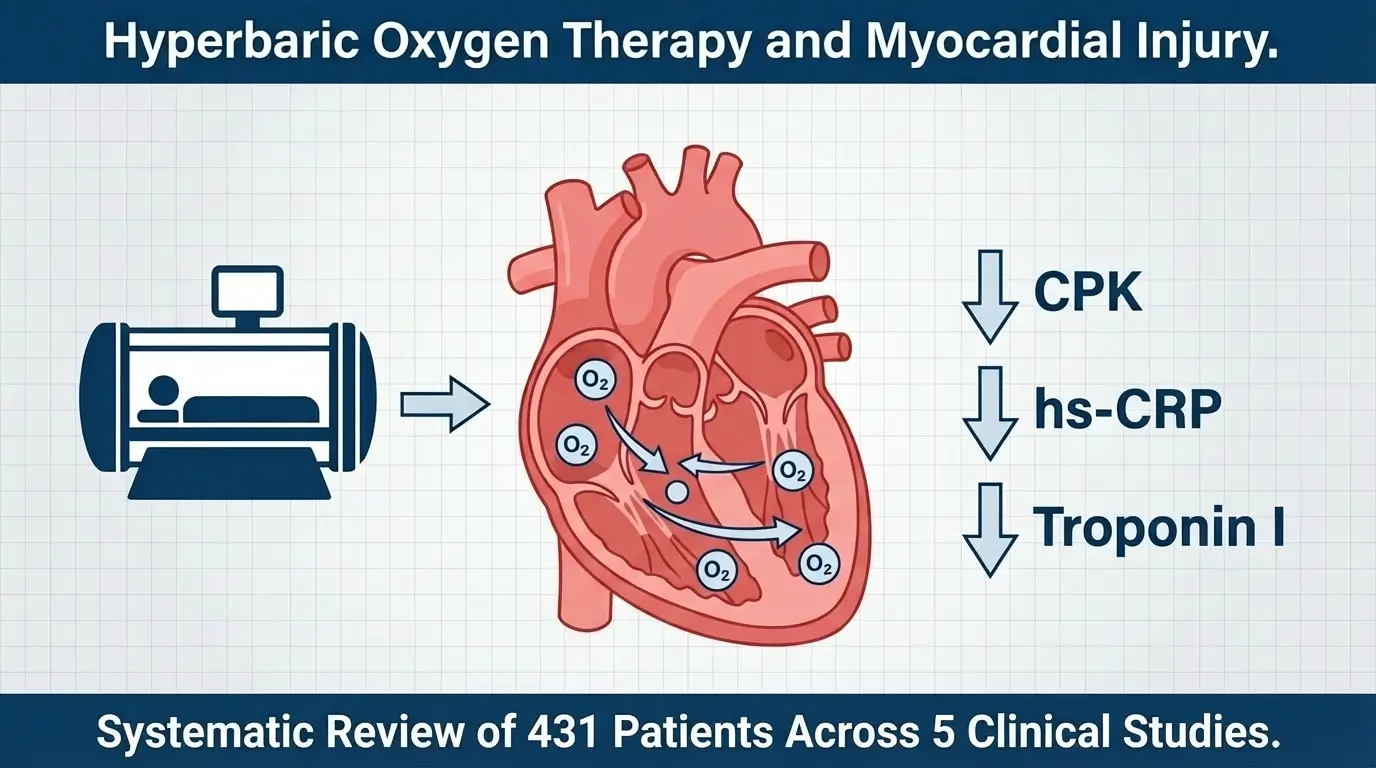
- A 2026 systematic review of five clinical studies involving 431 patients found that hyperbaric oxygen therapy (HBOT) reduced cardiac injury biomarkers by up to 35% and lowered inflammatory markers such as hs-CRP and endothelin-1.
- HBOT improved left ventricular ejection fraction in at least two trials, with one study showing LVEF rise from 46.27% to 50.81% over three weeks while control patients declined.
- A separate Cochrane review of six trials with 665 participants linked HBOT to a 42% reduced risk of death in acute coronary syndrome patients, though this was graded as low-quality evidence.
- The biomarkers affected by HBOT, including troponin, hs-CRP, and IL-6, are validated predictors of cardiac death, heart failure progression, and recurrent cardiovascular events.
- All major reviews concluded that small sample sizes, inconsistent HBOT protocols, and moderate risk of bias prevent clinical recommendations, and that large multicenter trials with long-term endpoints are needed.
A new systematic review published in Cureus analyzed five clinical studies involving 431 patients to assess whether hyperbaric oxygen therapy (HBOT) can reduce markers of heart muscle damage and inflammation.
The findings showed statistically significant reductions in several cardiac biomarkers, including a 35% drop in creatine phosphokinase (CPK) levels in one trial. Researchers also observed lower levels of inflammatory markers such as high-sensitivity C-reactive protein (hs-CRP) and endothelin-1. HBOT improved left ventricular ejection fraction (LVEF) in at least two of the included studies.
However, the review stopped short of recommending HBOT for routine cardiac care, citing small sample sizes, inconsistent protocols, and moderate risk of bias across the studies.
A separate Cochrane review of six trials with 665 participants found that patients with acute coronary syndrome who received HBOT had a 42% reduced risk of death (RR: 0.58, 95% CI 0.36 to 0.92), though this was graded as low-quality evidence. HBOT was also linked to faster pain relief and fewer major adverse cardiac events.
The Cochrane authors concluded that “the routine application of HBOT to these patients cannot be justified” based on current data.
A 2025 review in Medical Gas Research covering 78 studies echoed this position, finding that HBOT “demonstrates multifaceted benefits in the management of cardiovascular diseases” but that “its widespread adoption in clinical practice remains constrained by the lack of standardized treatment protocols and robust clinical evidence.”
A January 2025 review on HBOT in cardiovascular surgery published in the same journal found that preconditioning with HBOT reduced hospital stays, bleeding volumes, and postoperative complications in patients undergoing coronary artery bypass grafting.
What Is HBOT and How Does It Work on the Heart?
HBOT delivers 100% oxygen at pressures between 2.0 and 3.0 atmospheres absolute (ATA). This increases the amount of oxygen dissolved in blood plasma, independent of hemoglobin. The result is that more oxygen reaches ischemic (oxygen-starved) tissues, including damaged heart muscle.
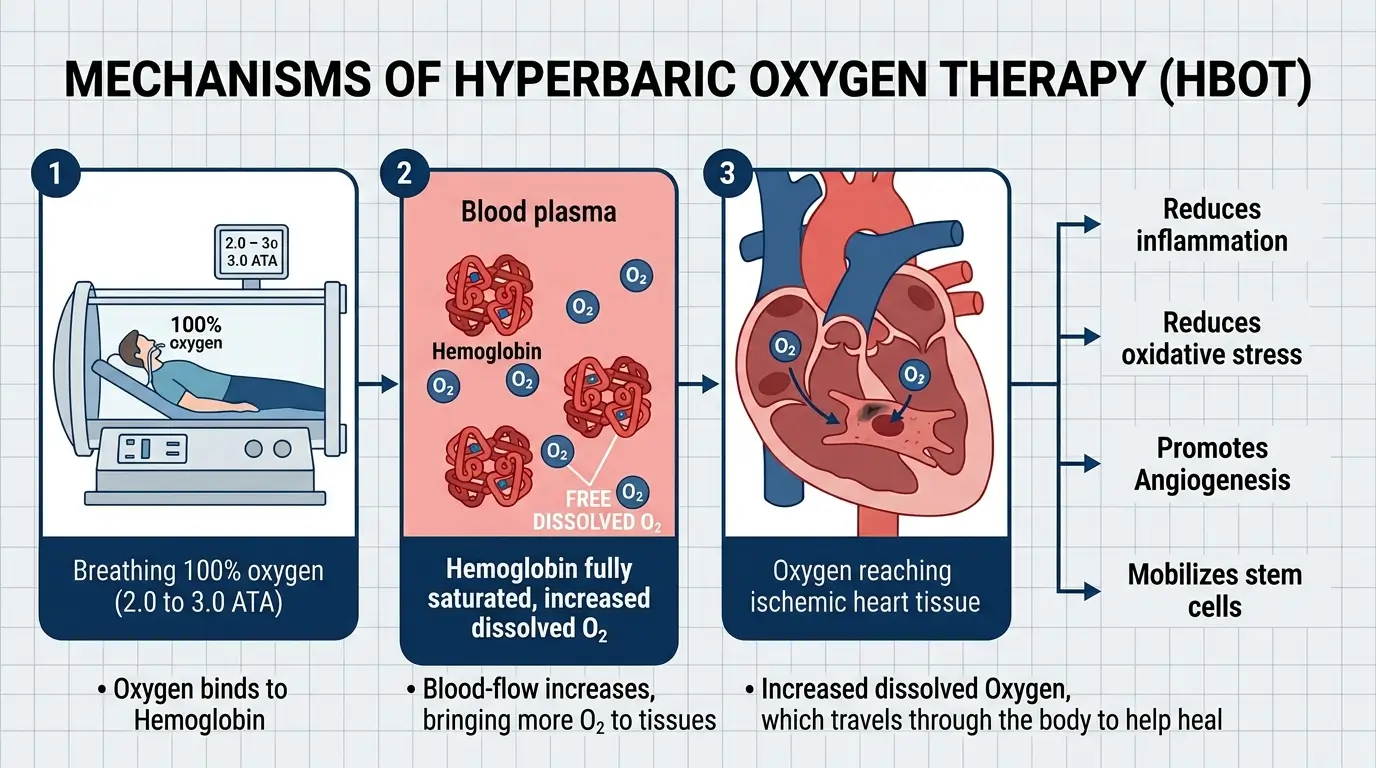
At a biological level, the Cureus review explains that HBOT works through several pathways. It suppresses inflammatory cytokine production by inhibiting NF-kB signaling. It reduces neutrophil adhesion to blood vessel walls, which limits the tissue damage that occurs during reperfusion (when blood flow returns after a blockage). It also supports mitochondrial function and reduces oxidative stress.
These mechanisms are relevant because inflammation and oxygen deprivation are two primary drivers of heart muscle death during and after a heart attack.
The Medical Gas Research review adds that HBOT promotes angiogenesis (growth of new blood vessels) in damaged heart tissue by upregulating a molecule called MALAT1, which suppresses the anti-angiogenic microRNA miR-92a.
In animal models, HBOT at 2.0 ATA for 60 minutes daily over 14 consecutive days reduced infarct size, lowered oxidative stress markers, and improved left ventricular systolic pressure.
What the Five Studies Found
The Cureus review included four randomized controlled trials and one observational study, conducted between 1997 and 2018. They covered three distinct cardiac populations: patients with acute myocardial infarction (AMI) receiving thrombolysis, patients undergoing elective coronary artery bypass grafting (CABG), and patients with chronic coronary artery disease (CAD) who had received drug-eluting stents.
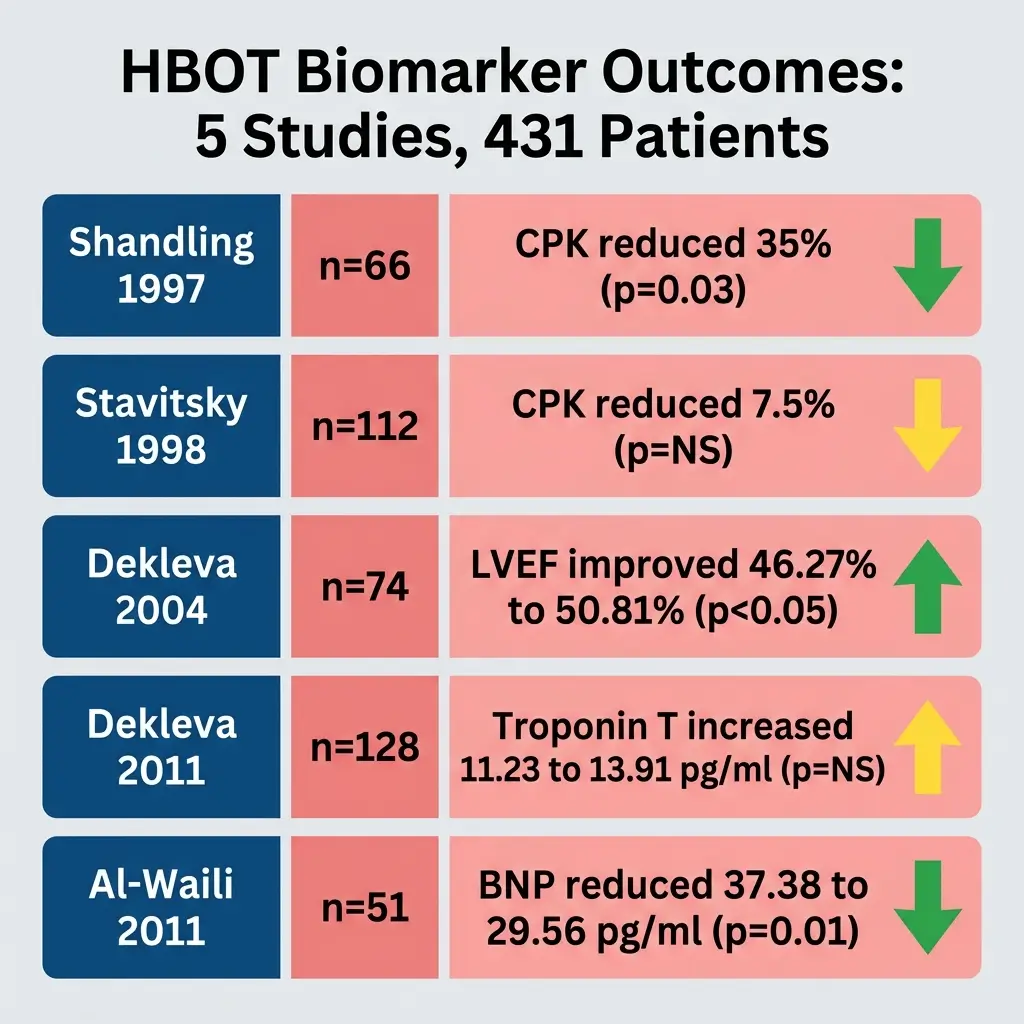
In the pilot HOT MI trial (Shandling et al., 1997, n=66), HBOT patients showed approximately 35% lower CPK levels at 12 to 24 hours post-treatment compared to controls (p=0.03). Their discharge LVEF was higher at 52.4% versus 47.3%, though this difference did not reach statistical significance.
The larger multicenter HOT MI trial (Stavitsky et al., 1998, n=112) found only a 7.5% CPK reduction with HBOT, and the result was not statistically significant.
Dekleva et al. (2004, n=74) reported more compelling results. Patients in the HBOT group saw their LVEF improve from 46.27% to 50.81% over three weeks. Control patients declined from 45.54% to 44.05%. This difference was statistically significant (p<0.05). Peak CPK was 35.3% lower in the HBOT group.
Alex et al. (2005, n=64) tested HBOT preconditioning before elective CABG. The double-blind trial found that patients who received two 30-minute sessions at 2.4 ATA before surgery had significantly lower postoperative troponin I levels (p<0.05). They also showed lower levels of soluble E-selectin, CD18, and heat shock protein 70, all markers of endothelial activation and cellular stress.
(Troponin I is the gold-standard biomarker for myocardial injury, making this finding particularly relevant to the question of whether HBOT provides real cardioprotection.)
Li et al. (2018, n=115) studied chronic CAD patients after drug-eluting stent placement. Patients received HBOT at 2.0 ATA for 80 minutes per session, six times per week, for four weeks.
The treatment group showed significant reductions in hs-CRP and endothelin-1 and an increase in nitric oxide (all p<0.05), suggesting improved endothelial function and reduced systemic inflammation.
Why These Biomarkers Matter
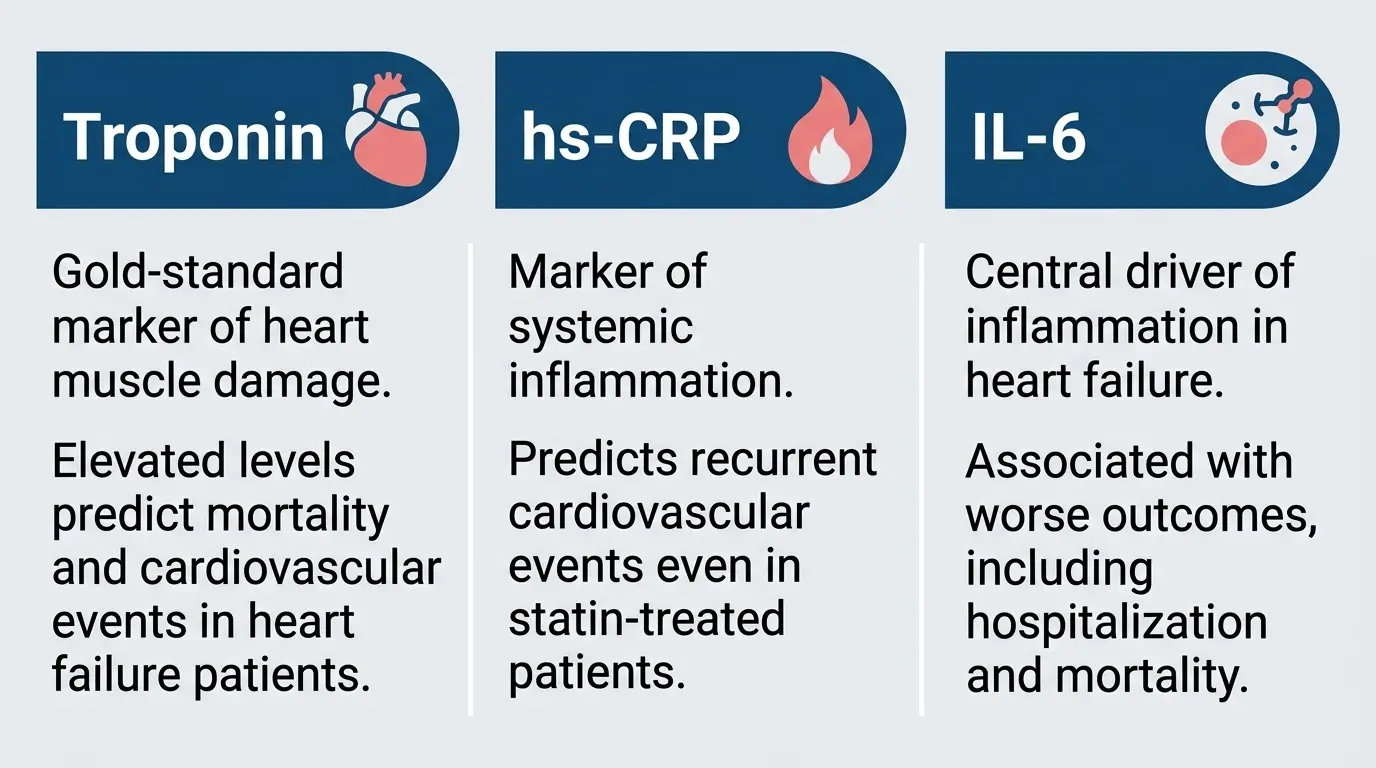
The review emphasizes that the biomarkers affected by HBOT have well-established prognostic value. Elevated high-sensitivity troponin T is independently associated with increased mortality and cardiovascular events in patients with chronic heart failure, according to an individual patient data meta-analysis cited in the review.
High-sensitivity CRP remains a validated marker of residual inflammatory risk even in statin-treated patients. Elevated interleukin-6 levels after ST-elevation myocardial infarction (STEMI) are associated with larger infarct size, reduced LVEF, and worse long-term outcomes.
A 2024 study published in JAMA analyzing prognostic cardiovascular biomarkers in the general population confirmed that these markers are “strongly associated with fatal and nonfatal cardiovascular events and mortality.” If HBOT can reliably lower these markers, the clinical implications could extend beyond biochemistry into actual patient outcomes.
The Limits of Current Evidence
The Cureus review is transparent about serious methodological constraints. Only five studies qualified out of 412 initial records screened.
The sample sizes ranged from 64 to 115 patients per study. HBOT protocols varied widely, from single preoperative sessions at 2.4 ATA to four-week courses at 2.0 ATA. Biomarker measurement timing and assay methods also differed across studies.
A post-hoc power analysis of the largest trial’s primary outcome (CPK reduction, n=66) yielded only 70% statistical power, below the conventional 80 to 90% threshold needed for clinical recommendations.
The researchers could not perform a meta-analysis because of incomplete reporting of dispersion measures (standard deviations) and substantial clinical heterogeneity. Several trials showed “some concerns” in risk of bias assessments using the RoB 2.0 tool, primarily related to allocation concealment and incomplete outcome reporting.
The Cochrane review faced similar issues. Its authors noted that “only two reported randomization procedures in detail and in only one trial was allocation concealment.” Most studies did not blind patients or researchers, raising the possibility of placebo effects.
The cardiovascular surgery review published in January 2025 acknowledged that “there remains a notable scarcity of literature specifically addressing its application within this field” and that “current studies may not be able to provide comprehensive data regarding dosage protocols, timing of administration and long-term effects.”

Safety Profile
HBOT appears to be well tolerated in cardiac patients based on the available data. The Cureus review cites a barotrauma rate of 1 to 2%. The Cochrane review reported no cases of neurological oxygen toxicity across its six trials. One patient experienced tympanic membrane damage.
Claustrophobia was an issue in single-person chambers, with one trial reporting a 15% incidence. (Ed. note: This safety profile is reassuring, but it applies to relatively short-term use in controlled settings, and the data on HBOT in patients with severe heart failure is limited.)
A review by Schiavo et al., cited in the Medical Gas Research paper, tracked 23 heart failure patients through an average of 39 HBOT sessions. Two cases of pulmonary edema occurred, both of which resolved with diuretic treatment.
The authors noted that HBOT-induced vasoconstriction increases venous return and arterial resistance, which can stress an already-compromised heart.
What Comes Next
All three major reviews call for the same thing: large-scale, well-designed prospective trials with standardized HBOT protocols, consistent biomarker measurement, and long-term clinical endpoints.
The Cureus authors write that “heterogeneity, methodological limitations, and small sample sizes prevent definitive clinical recommendations.”
The Cochrane review states that “an appropriately powered trial of high methodological rigour is justified to define those patients (if any) who can be expected to derive most benefit.”
The Medical Gas Research review suggests future work should focus on “identifying the optimal timing for HBOT intervention, refining treatment regimens, and investigating its synergistic effects when combined with conventional therapies.”
The biological rationale for HBOT in cardiac care is strong. The biomarker data, while limited, are largely consistent in the direction of benefit. The missing piece is scale.
Until a large multicenter trial demonstrates that these biochemical improvements translate into fewer heart attacks, fewer deaths, or better cardiac function over months and years, HBOT will remain an intriguing adjunct therapy without a firm foothold in cardiology guidelines.
References
- Cureus: Effects of Hyperbaric Oxygen Therapy on Myocardial Injury and Inflammatory Biomarkers: A Systematic Review With Qualitative Synthesis
- Cochrane Library: Hyperbaric oxygen therapy for acute coronary syndrome (Bennett MH et al., 2015)
- Cochrane Evidence Summary: Does hyperbaric oxygen therapy improve outcome after heart attack?
- Medical Gas Research: Application and progress of hyperbaric oxygen therapy in cardiovascular diseases (2025)
- Medical Gas Research: Hyperbaric oxygen therapy for cardiovascular surgery (2025)
- JAMA: Prognostic Value of Cardiovascular Biomarkers in the Population (2024)
- Nature Scientific Reports: The effect of hyperbaric oxygen therapy on myocardial function in post-COVID-19 patients (2023)