The FDA does not approve ozone therapy for any medical condition. This case report, published in the journal Undersea & Hyperbaric Medicine, shows why that matters.
What Happened to the Patient
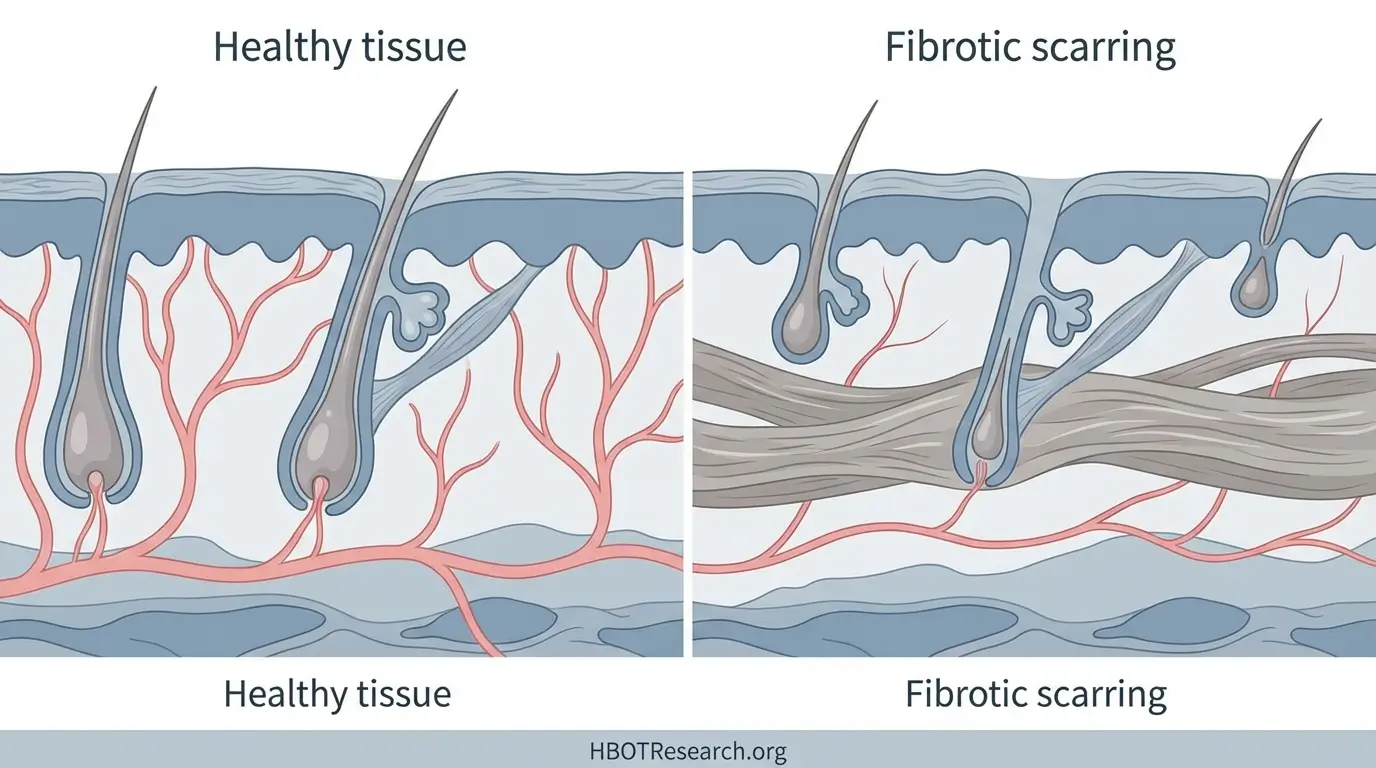
Medical ozone therapy involves injecting ozone gas into the skin or body. Practitioners promote it for infections, inflammation, and cosmetic purposes like collagen formation and hair growth. The evidence for these claims remains limited. The risks, however, are real.
The patient received ozone injections to her temples and central forehead from a dermatologist. The goal was to enhance hair growth and collagen formation. After the procedure, she developed worsening alopecia and scarring in the treated areas, the opposite of what she wanted.
A plastic and reconstructive surgeon eventually examined her. During surgery, the surgeon found significantly fibrotic, scarred, and poorly vascularized tissue. This discovery raised concerns about poor wound healing. The tissue damage was extensive enough that simple surgical repair would not suffice.
How Hyperbaric Oxygen Therapy Repaired the Damage
Her plastic surgeon referred her for hyperbaric medicine evaluation. The diagnosis: presumed skin flap vascular compromise from the ozone therapy.
She completed 40 hyperbaric oxygen treatments before undergoing scar tissue excision and local tissue rearrangement. The final result was acceptable cosmesis, according to the case report authors Patricia McCray and Kelly Johnson-Arbor.
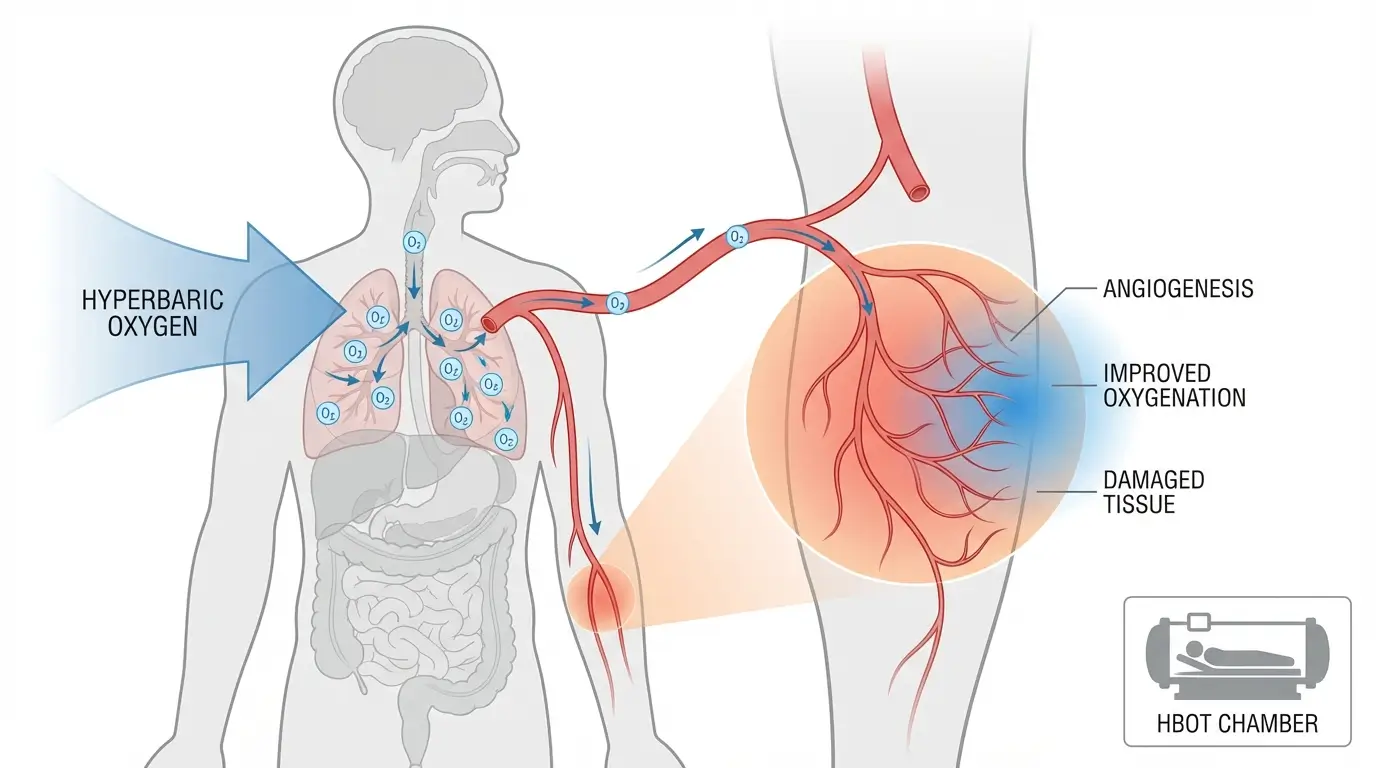
The treatment course illustrates the serious nature of these complications. Hyperbaric oxygen therapy (HBOT) involves breathing 100% oxygen at pressures higher than normal atmospheric pressure, typically 2.0 to 2.5 atmospheres absolute. Sessions last 90 to 120 minutes. At 40 sessions, this patient spent approximately 60 to 80 hours in a pressurized chamber.
(Ed. note: 40 HBOT sessions represents a significant treatment burden. Standard protocols for compromised tissue typically involve 20 to 30 treatments.)
HBOT works by increasing oxygen delivery to damaged tissues. According to the Undersea and Hyperbaric Medical Society, the therapy increases the oxygen-carrying capacity of plasma and promotes new blood vessel growth. For compromised skin flaps, this can mean the difference between tissue survival and tissue death.
The science behind HBOT for compromised grafts and flaps is well established. A 2017 review in Advances in Wound Care found that HBOT increases wound oxygen tension, stimulates collagen synthesis, and improves vascularization. Clinical studies show salvage rates of 75% to 90% for compromised flaps and grafts.
Why the FDA Does Not Approve Ozone Therapy
Ozone therapy lacks similar evidence. The U.S. Food and Drug Administration has not authorized or regulated ozone therapy for any medical purpose. The agency’s position is clear: ozone is a toxic gas with no known useful medical application.
The FDA states that ozone must be present in high concentrations to act as a germicide, far beyond what is safe for human exposure. Known risks include gas embolism, blood clots, and death.
What the Research Shows About Ozone Therapy Risks
Pulmonologist Vickram Tejwani of Cleveland Clinic put it this way: the possible side effects outweigh the potential health benefits. Tejwani noted that ozone therapy has not been studied in large-scale clinical trials conducted on humans.
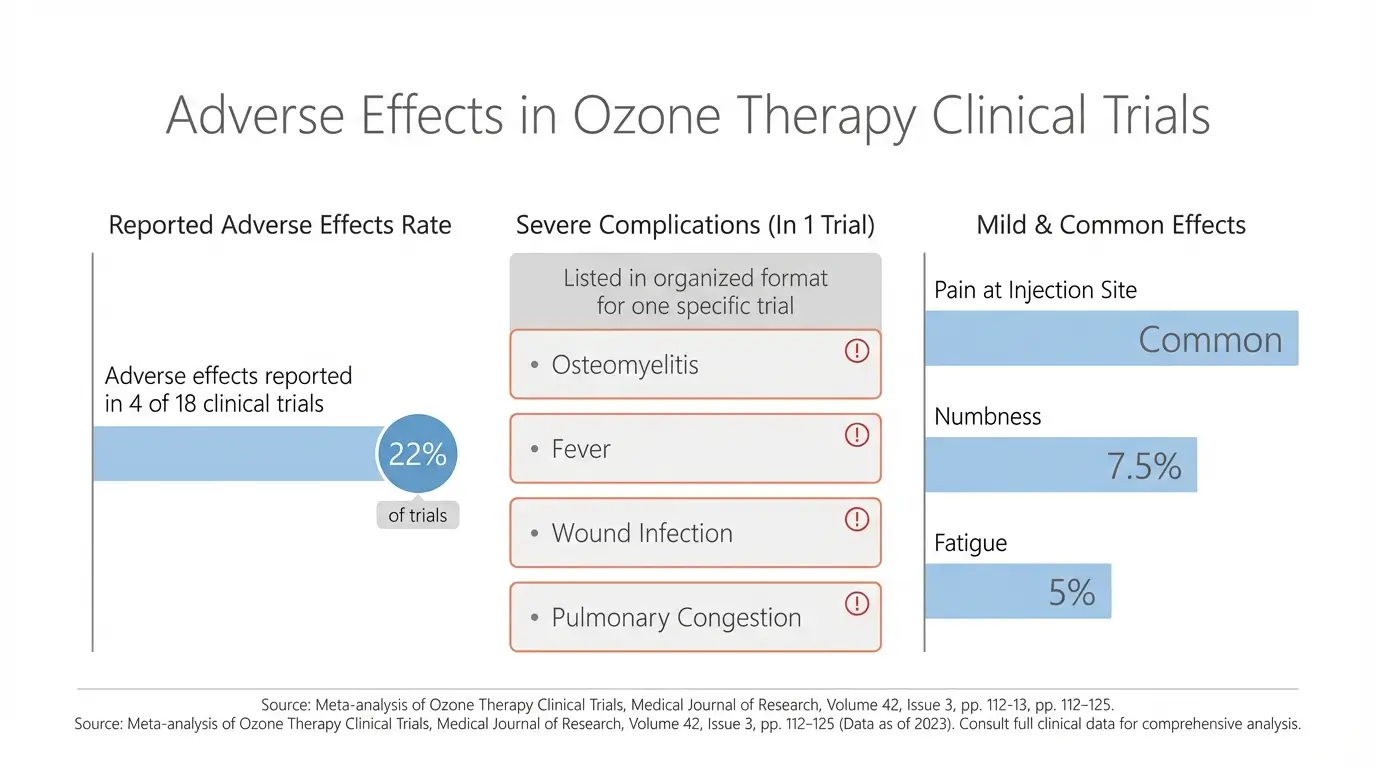
A 2022 systematic review in the Indian Journal of Dermatology examined 18 clinical trials involving ozone therapy for skin conditions. Researchers found that adverse effects occurred in 4 of 18 trials. One trial reported severe complications including osteomyelitis, fever, wound infection, and pulmonary congestion in five patients.
The review authors concluded that while ozone therapy showed some effectiveness for certain conditions, a greater number of high-quality clinical trials is needed to clearly establish safety. They specifically noted that health professionals must evaluate the risk-benefit ratio.
For cosmetic applications like the one in this case report, the evidence is even thinner. Some practitioners inject ozone into the scalp for hair growth. If excessive ozone is injected, it can be toxic and damage hair follicles rather than strengthen them, according to dermatology sources.
The case report authors stated their conclusion directly: while medical ozone therapy may result in favorable effects on a cellular level, the treatment is associated with significant clinical risks that may outweigh its potential therapeutic benefits.
They also highlighted a potential role for HBOT in treating ozone therapy complications. Systemic hyperbaric oxygenation may enhance vascularity and improve the quality of devitalized tissue in areas previously treated with medical ozone therapy, they wrote.
This creates a troubling situation. An unproven, unregulated therapy causes harm. The treatment for that harm requires 40 sessions of a proven therapy. The patient ends up in a hyperbaric chamber because of a cosmetic procedure that the FDA considers unsafe.
The regulatory gap is wide. Ozone therapy is not FDA approved, but practitioners still offer it. At least 13 U.S. states allow medical doctors to use ozone as an alternative medication, according to the Alabama Board of Medical Examiners. The treatment exists in a gray zone between regulation and practice.
What Patients Should Know Before Trying Ozone Therapy
Patients considering ozone therapy should know what they are getting into. The FDA has issued warnings. The evidence base is limited. Complications can require extensive treatment to repair. This case report documents what can happen when things go wrong.
The good news from this case: HBOT worked. The patient achieved acceptable cosmesis after treatment. The bad news: she needed 40 sessions and two surgeries to get there. The tissue damage from a procedure meant to help her hair and skin required months of specialized medical care.
For patients considering cosmetic procedures, the message is straightforward. Ask about FDA approval status. Ask about the evidence base. Ask what happens if something goes wrong. This case shows that ozone therapy complications can be serious and the recovery path can be long.
References
- Dermatologic medical ozone therapy complications treated with hyperbaric oxygen. Undersea & Hyperbaric Medicine. 2025;52(4):515-519.
- Hyperbaric Therapy for Skin Grafts and Flaps. StatPearls, National Library of Medicine.
- Hyperbaric Oxygen Therapy for the Compromised Graft or Flap. Advances in Wound Care. 2017;6(1):23-32.
- Effectiveness and Safety of Ozone Therapy for Dermatological Disorders. Indian Journal of Dermatology. 2022;67(4):479.
- Ozone Therapy: What It Is, Uses and Side Effects. Cleveland Clinic. December 21, 2022.
- FDA 21 CFR 801.415: Maximum acceptable level of ozone. U.S. Food and Drug Administration.