- Combining hyperbaric oxygen therapy with exosomes can significantly enhance tissue repair across multiple conditions.
- The combination works better than either treatment alone for neurological disorders.
- Animal studies show synergistic anti-inflammatory and neuroprotective effects.
- Researchers identify multiple mechanisms explaining the enhanced healing response.
- Clinical translation faces challenges including delivery optimization and standardization.
Researchers have identified a potential breakthrough in tissue repair by combining two distinct therapeutic approaches: hyperbaric oxygen therapy and exosomes. A comprehensive review published in Frontiers in Bioengineering and Biotechnology examines the synergistic effects of this combination across multiple disease models, revealing promising results that surpass either treatment used alone.
The research team, led by investigators from several Chinese medical institutions, analyzed existing studies on diabetic wounds, traumatic spinal cord injuries, peripheral nerve damage, and vascular diseases. Their findings suggest that pairing these therapies produces stronger anti-inflammatory, antioxidant, and tissue regeneration outcomes compared to single-treatment approaches.
How the Combination Works
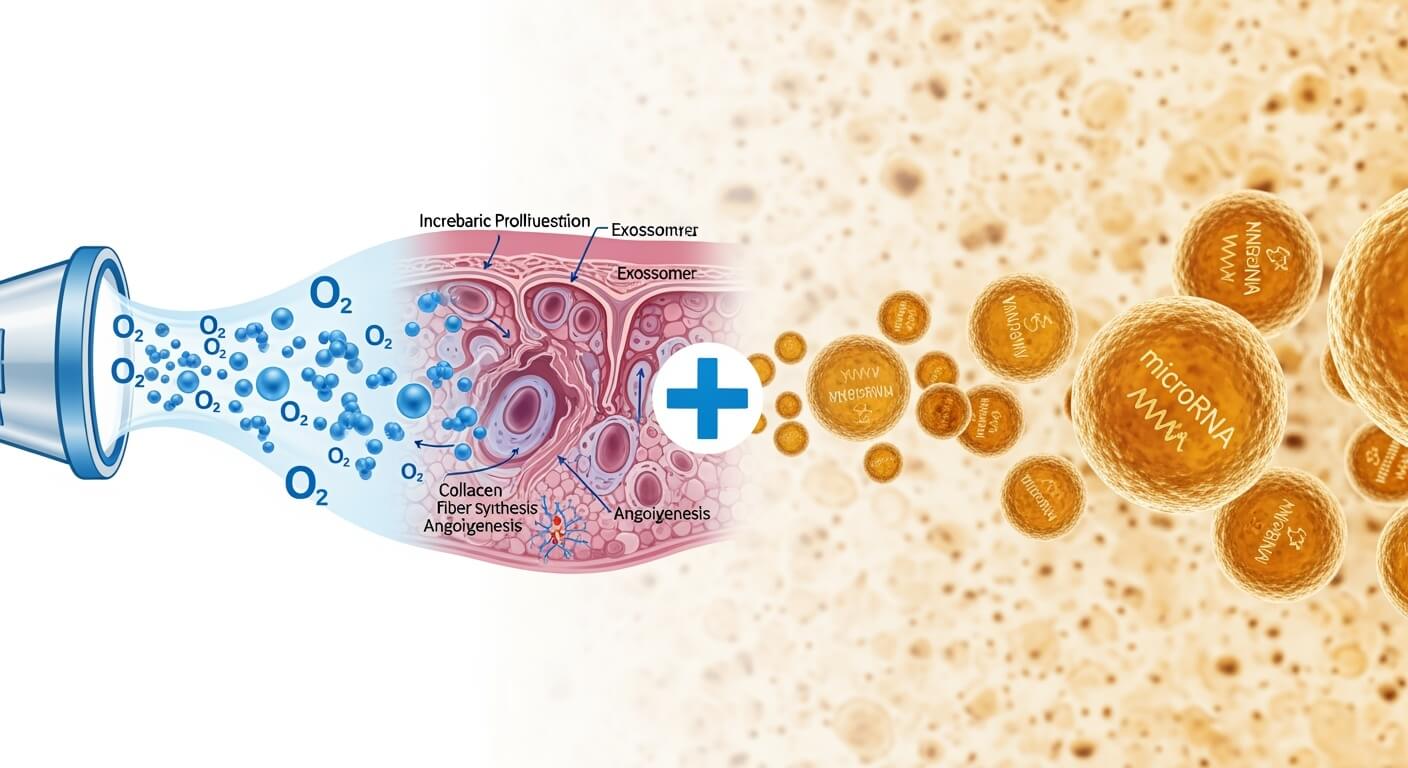
Hyperbaric oxygen therapy delivers pure oxygen at pressures greater than normal atmospheric levels, increasing dissolved oxygen in blood and tissues. This enhanced oxygenation promotes angiogenesis, boosts cellular metabolism, and reduces inflammation. The therapy has been endorsed by the International Society of Underwater and Hyperbaric Medicine for 14 clinical conditions, including refractory wounds and thermal burns.
Exosomes are nanoscale vesicles released by cells that carry proteins, RNAs, and other bioactive molecules. Those derived from mesenchymal stem cells have demonstrated tissue repair capabilities by modulating immune responses, enhancing cell proliferation, and facilitating wound closure. They offer advantages over stem cell therapy, including simpler structure, higher stability, easier storage, and lower immunogenicity.
The combination creates unexpected biological interactions. Hyperbaric oxygen modifies both the quantity and content of exosomes released from cells. Studies on human coronary artery endothelial cells exposed to hyperbaric oxygen showed increased expression of MALAT1, a long non-coding RNA that promotes angiogenesis. The treatment also downregulated inflammation-related genes in exosomes, including TLR5, FAAH2, and SLC11A1.
This phenomenon stems from what researchers call the “hyperoxia-hypoxia paradox.” While hyperbaric oxygen provides elevated oxygen levels, repeated exposure followed by return to normal conditions creates a state of relative hypoxia. This activates hypoxia-inducible factor-1α (HIF-1α), a protein that stabilizes under low oxygen and triggers exosome release. Clinical evidence supports this mechanism: patients with idiopathic acute sensorineural hearing loss showed dramatically increased serum exosome levels after hyperbaric oxygen treatment.
Exosomes also protect against hyperbaric oxygen-induced lung injury. Studies on bronchopulmonary dysplasia models showed that exosomes from mesenchymal stromal cells suppressed inflammatory responses and promoted tissue regeneration when administered alongside hyperbaric oxygen exposure. The exosomes reduced inflammatory lymphocytes and pro-inflammatory cytokines while facilitating alveolar epithelial cell proliferation.
Results Across Disease Models
The most extensive evidence comes from studies on diabetic wounds, which affect millions globally and frequently resist conventional treatment. Research by Yin and colleagues demonstrated that exosomes from human umbilical vein endothelial cells exposed to hyperbaric oxygen promoted browning of white adipose tissue. These beige adipocytes induced anti-inflammatory M2 polarization in macrophages and enhanced fibroblast proliferation, accelerating wound closure.
Ed. note: The diabetic wound findings represent in vitro observations that require validation in animal models and human trials.
Traumatic spinal cord injury studies provide the strongest evidence for clinical potential. In the first investigation of this combination, Cheshmi and colleagues treated rats with exosomes from human placental mesenchymal stem cells plus hyperbaric oxygen at 2.5 atmospheres for 90 minutes across three sessions.
Compared to either treatment alone, the combination produced higher Basso-Beattie-Bresnehan locomotor scores on days 1, 3, 7, and 14 post-injury. The rats also performed better on narrow beam walking tests and showed significantly shorter electromyography latency.
A follow-up study by Hjazi and colleagues tested exosomes from human menstrual blood stem cells combined with hyperbaric oxygen in a similar injury model. These stem cells offer practical advantages: non-invasive collection, rapid proliferation with a 19.4-hour doubling time, and minimal immunogenicity.
The combination again outperformed single treatments, though some differences did not reach statistical significance, possibly due to small sample sizes or immune factors affecting exosome delivery in cross-species applications.
Spinal cord ischemia-reperfusion injury, which can occur after abdominal aortic surgery, showed similar benefits. Jafari and colleagues found that combining hyperbaric oxygen with placental stem cell exosomes markedly reduced motor deficit index scores at 6, 12, and 48 hours post-injury compared to single treatments. Neuronal density increased, oxidative stress decreased, and histopathological damage improved.
For peripheral nerve damage, Amiri and colleagues examined sciatic nerve injury in rats. The combination therapy produced larger nerve volume, thicker myelin sheaths, and more nerve fibers compared to either hyperbaric oxygen or exosomes alone. Behavioral assessments showed considerably reduced electromyography delay and elevated sciatic nerve function index scores across 28 days of observation.
Vascular disease models yielded encouraging results. In hind limb ischemia, hyperbaric oxygen-induced exosomes markedly enhanced blood flow. For myocardial infarction, co-administration substantially decreased infarct size and increased cardiac ejection fraction by an average of 14% compared to exosomes not exposed to hyperbaric conditions.
Mechanisms of Synergy
The combination produces three key synergistic effects. First, it amplifies anti-inflammatory responses beyond what either treatment achieves individually. Studies across multiple injury models showed that tumor necrosis factor-α and interleukin-1β levels dropped significantly more with combined treatment. The therapy also boosted anti-inflammatory interleukin-10 expression, which promotes neuronal survival after injury.
Second, the pairing enhances antioxidant capacity while suppressing oxidative stress. Multiple investigations documented elevated levels of glutathione, catalase, and superoxide dismutase alongside reduced reactive oxygen species when both therapies were applied together. This balanced response helps prevent the cellular damage that slows tissue repair.
Third, the combination provides superior neuroprotection. Stereological measurements showed that total spinal cord volume in combined treatment groups reached approximately double that of control groups. The effect stemmed partly from reduced activity of caspase-3, an apoptosis-related protein. Lower caspase-3 activity translated to enhanced neuronal survival and proliferation across injury sites.
Safety Profile and Challenges
A systematic review and meta-analysis examining 24 randomized controlled trials with 1,497 subjects found an overall adverse effect rate of 30.11% for hyperbaric oxygen therapy, with ear discomfort being most common. When treatment protocols limited sessions to 10 or fewer and chamber pressure stayed below 2.5 atmospheres, adverse effects decreased markedly.
Exosomes from autologous cells show good tolerance, but allogeneic or xenogeneic sources may trigger immune rejection or allergic reactions. Surface proteins including MHC complexes can activate T cell responses. Contamination during extraction and purification presents infection risks. The hyperbaric environment itself may compromise exosomal membrane stability, releasing contents prematurely and activating unintended cellular signaling pathways.
Effective delivery remains a major bottleneck for clinical translation. Exogenous exosomes have short circulation times and face clearance by macrophages. The hyperoxic environment created by hyperbaric oxygen may oxidize and degrade exosomal lipid membranes, reducing bioavailability. Production costs, quality assurance, and standardized extraction and storage techniques represent practical challenges requiring resolution.
Path to Clinical Application
No clinical trials combining hyperbaric oxygen with exosomes have been conducted in humans. While hyperbaric oxygen therapy has extensive clinical use for traumatic conditions, the addition of exosomes requires substantial investigation to guarantee safety and efficacy.
Future research should examine various treatment protocols to identify optimal combinations and dosages. Randomized controlled trials need to assess long-term efficacy and safety, including tissue healing rates, inflammatory marker levels, and adverse event occurrences.
Multi-omics approaches using transcriptomics and proteomics may clarify the molecular interactions between hyperbaric oxygen and exosomes, revealing potential synergistic pathways and informing new intervention techniques. Given the heterogeneity of exosomes, subsequent research should investigate ideal ratios of distinct exosome sources to hyperbaric oxygen exposure. This could enhance treatment specificity and potentially reduce allogeneic rejection risks.
Standardization presents another critical challenge. Production costs, quality assurance, and standardized extraction and storage techniques require resolution before widespread clinical implementation. Establishing international consensus guidelines will ensure clinical trials adhere to Good Clinical Practice standards and facilitate uniform implementation globally.
Advances in bioengineering and nanotechnology may enhance exosome delivery systems. Integrating targeting ligands or protective coatings could improve stability in hyperbaric environments. Identification and validation of biomarkers would enable response monitoring and personalized treatment approaches.
Despite minimal toxicity in animal trials, potential human risks require careful evaluation. The transition from rodent models to large animal studies, then to human trials, will test whether the synergistic effects observed translate across species and physiological scales.
The research suggests a promising avenue for treating conditions that currently have limited options. The combination leverages complementary mechanisms: hyperbaric oxygen enhances the cellular environment for healing while also modifying exosomes to make them more therapeutically potent; exosomes deliver targeted repair signals while protecting tissues from oxygen toxicity. This bidirectional interaction may explain the superior outcomes compared to either treatment alone.
For patients with diabetic wounds, spinal cord injuries, and vascular diseases, this approach could represent a meaningful advance. The therapy’s reliance on established hyperbaric oxygen infrastructure combined with cell-free exosome products may facilitate adoption once safety and efficacy are confirmed.
The next decade will likely see expanded investigation of this combination across additional conditions and the development of standardized protocols for clinical use. Treatment strategies based on personalized medicine will customize optimal plans for patients with various diseases, matching exosome sources and hyperbaric oxygen parameters to individual needs.
References
- Frontiers in Bioengineering and Biotechnology – Hyperbaric oxygen in combination with exosomes: a new strategy to promote tissue repair
- PubMed Central – Hyperbaric oxygen in combination with exosomes: a new strategy to promote tissue repair
- Stem Cell Research & Therapy – Stem cell-derived exosomes: emerging therapeutic opportunities for wound healing
- Nature – Clinical applications of stem cell-derived exosomes
- Frontiers in Cell and Developmental Biology – Bioengineered MSC-derived exosomes in skin wound repair and regeneration
- Stem Cell Research & Therapy – Therapeutic potential of mesenchymal stem cell-derived exosomes in wound healing